In a major acceleration of bird flu pandemic orchestration and mass vaccination infrastructure, Moderna, Inc. yesterday announced that “the first participants in the U.S. and UK have been dosed in a Phase 3 study of the Company’s investigational mRNA-based H5 pandemic influenza vaccine candidate, mRNA-1018.”
This is the first mRNA-based pandemic influenza (H5 bird flu) vaccine to reach Phase 3 clinical trials anywhere in the world.
The move comes after President Donald Trump selected a military influenza pandemic policy architect—Dr. Erica Schwartz, who built and enforced surveillance, vaccination, and compliance systems—to take control of the CDC.
Signalling its high confidence that there will be future demand for influenza vaccines, Moderna announced on the same day that another of its influenza jabs, mCOMBRIAX® (mRNA-1083), has been granted marketing authorization by the European Commission (EC).
Moreover, the Trump administration is currently funding gain-of-function experiments on influenza pathogens.
Just as coronavirus gain-of-function experiments and vaccine development (the problem and the solution) were simultaneously carried out before the COVID-19 pandemic, the same dual-track approach is now being applied to influenza—advancing high-risk pathogen work in parallel with the rapid rollout of mRNA vaccine platforms designed to respond to it.

Thousands to Be Injected
Moderna’s Tuesday press release reveals that 4,000 adults will be injected with the bird flu shot:
The Phase 3 study will evaluate the safety and immunogenicity of mRNA-1018 in healthy adults ages 18 years and older and is expected to enroll approximately 4,000 adults in the U.S. and UK. If successful, global regulatory submissions for mRNA-1018 will be further supported by positive data from the pivotal Phase 3 trial of Moderna’s investigational seasonal influenza vaccine, mRNA-1010, which has been accepted for review in the U.S., the European Union, Canada and Australia.
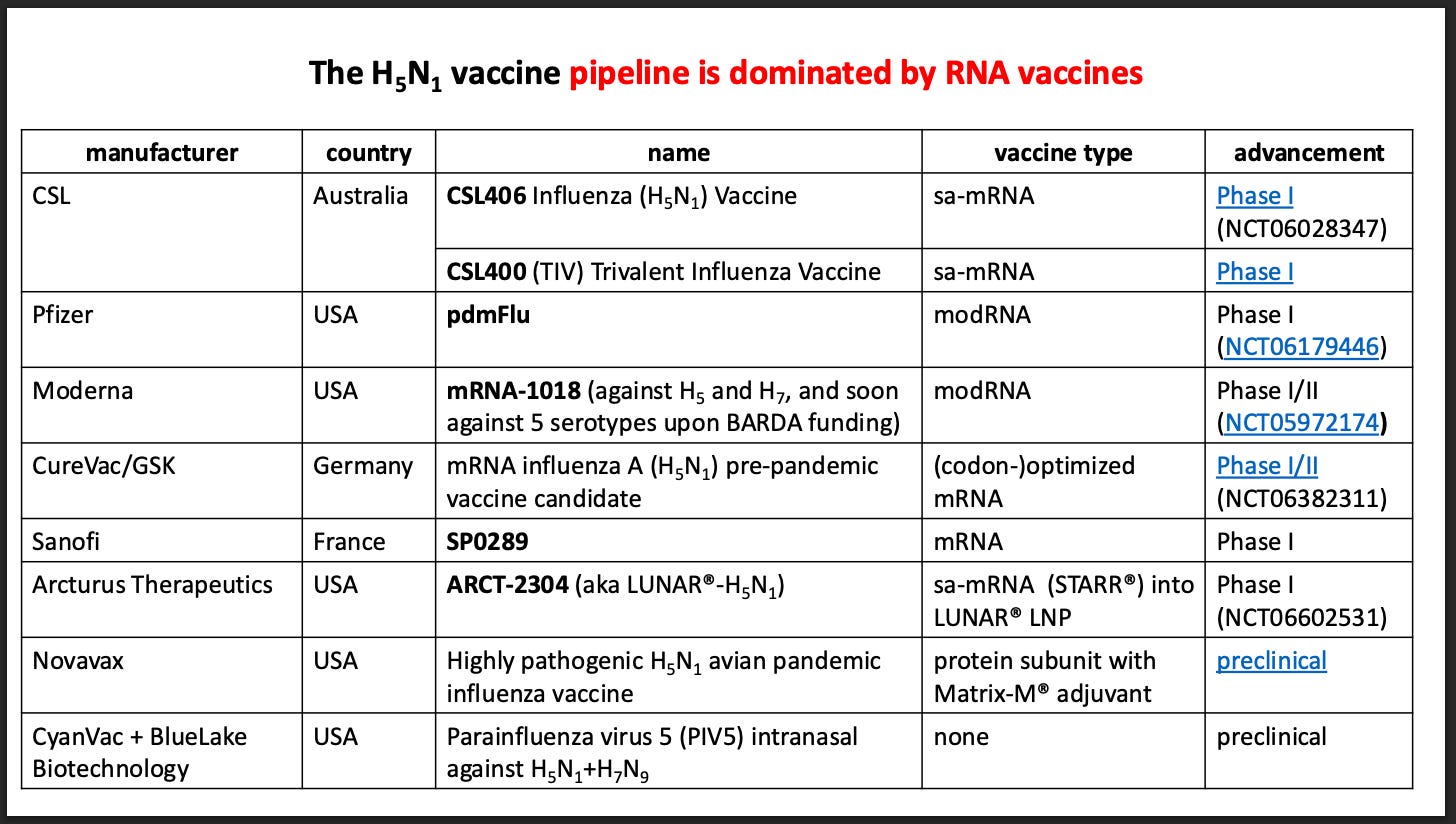
mRNA-1018 Resurrected
In December 2025, this website was the first to report that Bill Gates’ vaccine cartel, the Coalition for Epidemic Preparedness Innovations (CEPI), had invested $54.3 million to help advance mRNA-1018 to licensure.
CEPI’s massive contribution brought mRNA-1018 back to life, after HHS had terminated its multi-hundred-million-dollar commitment to Moderna for the vaccine.
mod?mRNA
The WHO confirms mRNA?1018 is classified as a “mod?mRNA” vaccine, which means it uses synthetic mRNA with nucleoside modifications (such as 1?methylpseudouridine) instead of unmodified, naturally occurring uridine.
N1-methylpseudouridine—the modified base used in mRNA vaccines—can induce ribosomal frameshifting, causing cells to produce unintended “rogue” proteins in roughly 8% of translation events and triggering immune responses to those proteins in about one-third of vaccinated individuals, introducing misdirected immune activity that experts from Oxford University and the University of Cambridge warn has “huge potential to be harmful.”
Moderna’s Bets on Bird Flu Pandemic
Moderna’s press release confirms the jab is for a coming avian influenza pandemic:
“H5 influenza, or bird flu, remains a pandemic threat. The start of our Phase 3 trial for an H5 influenza vaccine marks a significant milestone in our ongoing efforts to strengthen global pandemic preparedness,” said Stéphane Bancel, Chief Executive Officer of Moderna. “Our platform technology’s efficiency and scalability are critical to supporting global health security and responding to potential future threats.”
“The first-of-its-kind pivotal trial of an mRNA-based pandemic influenza vaccine underscores the potential of this technology to reshape how we confront emerging pathogens,” said Dr. Richard Hatchett, CEO of CEPI. “If successful, these efforts could transform our ability to respond swiftly and equitably to one of the world’s most enduring threats.”
…
“Although the current risk to the human population of avian influenza remains low, the virus continues to evolve and spread in birds and various animal hosts in the UK and elsewhere,” said Dr. Richard Pebody, Director of Epidemic and Emerging Infections at the UK Health Security Agency (UKHSA). “UKHSA remains alert to the potential that this pathogen could adapt to spread from person to person, and will continue to monitor all available data. This important initiative to trial a new mRNA pandemic influenza vaccine is a key step towards further strengthening our ability to protect people against future influenza pandemics.”
DARPA-Moderna Connection
Moderna’s mRNA development has been funded by the Defense Advanced Research Projects Agency (DARPA), the U.S. military’s advanced research agency.
In 2013, DARPA awarded Moderna up to about $25 million (including a $700,000 “seedling” grant) to develop mRNA vaccines.
In 2018, DARPA funded work tied to the EcoHealth DEFUSE project, which proposed collecting bat coronaviruses and genetically modifying them by inserting furin cleavage sites (FCS) and altering receptor-binding domains (RBD)—the defining features that set SARS-CoV-2 apart—while simultaneously developing mRNA vaccines to respond to those engineered threats before the pandemic began.
A later 2020 DARPA?Moderna agreement committed up to $56 million for a mobile manufacturing platform for more nucleic?acid vaccines.
Under its Pandemic Prevention Platform, DARPA contractors were said to be assembling influenza viruses from scratch using synthetic DNA fragments, describing a process where “overlapping oligonucleotides… [are] pooled, ligated and amplified… error corrected… assembled… and delivered… for virus rescue.”
This is a step-by-step system for constructing synthetic influenza viruses in the lab.
The same document warns that “seasonal or a weaponized, highly pathogenic, influenza strain remains a significant global challenge.”
Taken together, DARPA’s program outlines a single integrated pipeline to build or reconstruct the influenza virus, sequence it, and rapidly convert that sequence into an mRNA injection.
That same pipeline is now visible in real time with Moderna’s Phase 3 bird flu shot.
Bottom Line
Moderna’s Phase 3 bird flu injection is the real-time deployment of a DARPA-backed system that has long treated influenza viruses and mRNA countermeasures as part of the same pipeline—where pathogens can be constructed or reconstructed in the lab, converted into genetic sequences, and rapidly turned into injectables.
That system—funded by DARPA, revived by CEPI, and framed through ongoing pandemic warnings—is now moving into mass human trials, under a federal apparatus set to be led by the very architect of its military-run influenza pandemic surveillance, vaccination, and compliance infrastructure.
Your support is crucial in helping us defeat mass censorship. Please consider donating via Locals or check out our unique merch. Follow us on X @ModernityNews.
More news on our radar